Labs and Lytes 036
Author: Dr Renata Pajtak
Reviewers: Dr Greg Brogan, A/Prof Chris Nickson
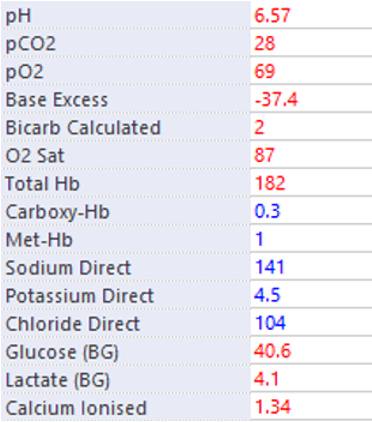
You are presented with the following arterial blood gas (ABG) result at the start of a shift:
NB. PO2 and PCO2 are measured in mmHg, Hb is measured in g/L, and electrolytes/ glucose/ lactate are measured in mmol/L
Q1. Describe and interpret the ABG
A suggested approach to basic ABG interpretation is as follows:
- Determine the change in pH, is it acidaemia or alkalaemia?
In this case the pH was 6.57, therefore the patient is severely acidaemic. - Determine the change in PCO2: is the PCO2 contributing to the change in pH or is it reacting to it
In this case the PCO2 was 28, indicating it is reactive - Determine the change in standard base excess: metabolic acidosis or alkalosis?
The base excess is -37.4 indicating a metabolic acidosis - Determine any compensation
PaCO2 = (1.5 × HCO3) + 8 for metabolic acidosis
Based on Winters formula, the PCO2 should be 11 in this case, with full respiratory compensation. As the PCO2 is 28, compensation is not complete.
This may be due to rapid onset of metabolic acidosis (respiratory compensation occurs within minutes but may not be maximal for 12 to 24h, or other factors limiting respiratory compensation (e.g. coexistent respiratory disease or decreased respiratory drive from decreased level of consciousness). - Determine the Anion Gap
Anion Gap = (Na+) – (Cl-+HCO3-)
141-(104+2)= 35 mmol/L
This indicates a high anion gap metabolic acidosis, as although normal anion gap varies with different assays, it is typically 4 to 12 mmol/L - Determine an Osmolar gap for high anion gap metabolic acidosis
Osmolar gap= measured osmolality – (2× Na+ + glucose + urea)
However, measured osmolality is not provided
The key findings on interpretation this ABG are:
- Severe acidaemia due to high anion gap metabolic acidosis with incomplete respiratory compensation
- Marked hyperglycaemia
- Hyperlactaemia (that doesn’t fully explain the high anion gap)
- Elevated hyperglycaemia (suggestive of marked dehydration in this case)
Q2. Should sodium be corrected for hyperglycaemia when calculating the anion gap?
No.
Sodium should only be corrected for hyperglycaemia when calculating free water deficit in hypernatraemia, not when calculating the anion gap:
“The anion gap reflects the balance between positively and negatively charged electrolytes in the extracellular fluid. Glucose is electrically neutral and does not directly alter the anion gap. However, glucose is osmotically active so water is pulled into the extracellular fluid. This has a dilutional effect on all extracellular electrolyte concentrations, both positive or negative, and so the anion gap is minimally altered.”
Dr Paul Young in LITFL’s Metabolic Muddle 005
Q3. What are the differential diagnoses for the metabolic disorder?
Diabetic Ketoacidosis (DKA) is the most likely major cause due to the presence of severe hyperglycaemia. Other causes of ketoacidosis include starvation ketoacidosis and alcoholic ketoacidosis.
Other causes of high anion gap include:
- Lactic acidosis
- Renal failure/ Uraemia
- Toxicants (such as methanol, ethylene glycol and other toxic alcohols, pyroglutamic acidosis, iron overdose, Salicylates)
If you like mnemonics, these can be arranged into the brief and easy mnemonic LTKR (“left total knee replacement”) or the more comprehensive/ complicated MUDPILES.
Q4. Which additional investigations could be performed to determine the underlying cause?
Key investigations to help confirm the underlying cause(s) include:
- Ketones (blood, urine)
- Urea, electrolytes, creatinine (UEC)
- Serum osmolality
Many other investigations may also be useful depending on the clinical context (e.g. ECG, CXR, serum bHCG).
Q5. What are the diagnostic criteria for diabetic ketoacidosis (DKA)?
There are no definitive criteria, but criteria similar to these are commonly used:
- pH <7.3
- ketosis (ketonemia or ketonuria)
- HCO3- < 18mmol/L due to high anion gap metabolic acidosis (HAGMA)
- Hyperglycaemia
- Anion gap >10 mmol/L
Note that euglycaemic DKA can occur… as can DKA with normal pH and HCO3 (if co-existent metabolic alkalosis from profuse vomiting, for instance)!
Q6. What management would you commence for DKA?
Goals of treatment include:
- Address immediate life-threats
- Correct dehydration
- Reverse ketosis, correct acidosis and glucose
- Monitor for complications of DKA and its treatment: cerebral oedema, hypo/hyperkalaemia, hypoglycaemia
- Identify and treat any precipitating cause
Initiate management in accordance with your hospital guideline for DKA, which will typically include therapies such as those described below.
Resuscitation to address immediate life-threats:
- Intubation for airway protection if required
- Oxygen to target SpO2 92-96%
- IV access
- Fluid boluses if required (e.g. hypovolemic shock)
Manage acid-base and electrolyte abnormalities
- Crystalloid fluid replacement (patients are usually markedly dehydrated)
- In adults a common regimen, that can be adjusted to the individual, is:
- 1L over 1h, then
- 1L over 2h, then
- 1L over 4h, then
- 1L every 6-8h until hydrated/drinking
- Slower rates are preferred in children due to higher risk of cerebral oedema
- Traditionally, normal saline was used, however increasing balance salt solutions are being used as they may lead to faster resolution of DKA (Self et al, 2020) and normal saline contributes to a normal gap metabolic acidosis.
- In adults a common regimen, that can be adjusted to the individual, is:
- Start insulin infusion at 0.1u/kg/h once fluid resuscitation commenced (and K >3.5 mmol/L)
- Aim to lower the glucose by 1-2mmol/L/h by adjusting insulin infusion rate to monitored blood glucse levels (refer to hospital guideline)
- Once glucose <15mmol/L provide dextrose 5% 100ml/ hr
- Correct osmolality by 3mosmol/kg/hr
- Continue insulin infusion until DKA resolved and patient is eating and drinking
- Long-acting insulin may be initiated while the insulin infusion is running once the patient is well hydrated
- Potassium (K) needs to be replaced one the serum potassium is < 5mmol/L
- E.g. 20 mmol/h if K <4 mmol/L or 10 mmol/h if K is between 4 and 5 mmol/L
Seek and treat underlying cause/ trigger
Supportive care and monitoring, including:
- Capillary blood glucose (initially q1h)
- Urinary catheter
- Monitor capillary ketones (initially q1h)
- Monitor blood gas (initially q2h)
- Neurological observation
- Cardiac monitoring (hyper/hypokalaemia)
Disposition
- Admit to ICU due to severe DKA
Q7. Why is it important to replace potassium even though it appears to be normal?
In DKA a patient can be potassium depleted whilst presenting with a hyperkalaemia. There are four main reasons for this:
- There is no insulin to activate the NA/K channel to drive potassium (K+) into cells, so it causes the extracellular [K+] to rise.
- There is a decreased response to catecholamines, so less K+ is driven into cells
- There is a metabolic acidosis therefore H+ ions are being moved into cells in exchange for K+ ions
- If the glucose concentration is so high that is causes a osmotic diuresis then water leaves the intracellular environment resulting in an increase in [K+] in the intracellular environment and consequently causing an efflux of K+ ions into the extracellular serum.
Once insulin therapy is commenced and acidaemia begins to resolve, the serum [K+] will fall as K+ moves back inside cells from the extracellular compartment.
References
- Alfred Health Guideline. Management of Diabetic Ketoacidosis in Adults. Accessed 9 Feb 2022.
- Nickson CP. Diabetic ketoacidosis. LITFL CCC. Accessed 9 Feb 2022. Available at URL: https://litfl.com/diabetic-ketoacidosis/
- Self WH, Evans CS, Jenkins CA, et al. Clinical Effects of Balanced Crystalloids vs Saline in Adults With Diabetic Ketoacidosis: A Subgroup Analysis of Cluster Randomized Clinical Trials. JAMA Netw Open. 2020;3(11):e2024596. Published 2020 Nov 2. doi:10.1001/jamanetworkopen.2020.24596 [Fulltext article]
- Young PJ. Hypernatraemia and acidosis. LITFL CCC. Accessed 9 Feb 2022. Available at URL: https://litfl.com/hypernatraemia-and-acidosis/
All case-based scenarios on INTENSIVE are fictional. They may include realistic non-identifiable clinical data and are derived from learning points taken from clinical practice. Clinical details are not those of any particular person; they are created to add educational value to the scenarios.