Labs and Lytes 041
Author: Dr Viveka Nainani
Reviewer: Dr Greg Brogan
You, as the ICU registrar, attended a “Code Blue” call for a 79-year-old man who has been in hospital for 2 months after coronary artery bypass graft (CABGx2) surgery that was complicated by tamponade, delirium, pulmonary embolism and slow respiratory wean (recent tracheostomy decannulation).
You noted the following findings on arrival:
- Patent airway
- Minimal respiratory effort
- SpO2 60% when breathing room air, improved to 99% on 15L/min O2 via non rebreather mask (NRM)
- BP 110/70 mmHg, SR 90/min
- GCS 3, PEARL 3mm
You were also shown an arterial blood gas (ABG) result, that had just been performed:
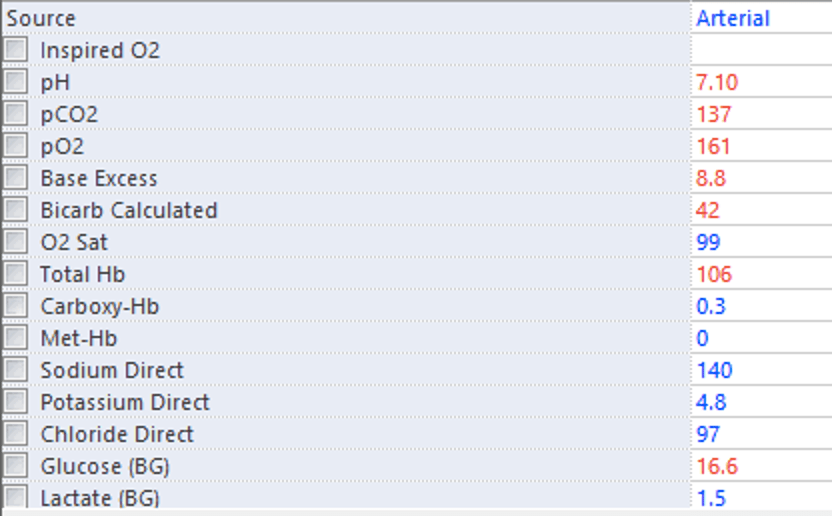
Q1. What are the key findings on the ABG? (Taken while on 15L/min O2 via non rebreather mask)
The key finding is Type 2 respiratory failure (marked hypercapnoea with PaCO2 = 137 mmHg).
This has resulted in a respiratory acidaemia. The patient is also hypergycaemic (16.6 mmol/L; most likely due to a stress response or due to pre-exisiting diabetes mellitus) and mildly anaemic (Hb 106 g/L).
This patient suffered a hypercapnoeic respiratory arrest in setting of hypoventilation, likely due to a central cause (e.g. brainstem stroke).
Q2. What is the A-a gradient?
The A-a gradient is the difference between the alveolar pO2 (derived from the alveolar gas equation) and arterial pO2, in mmHg. A normal A-a gradient is between 5-10 mmHg, however this is increased with age. For a 79-year-old male the normal A-a gradient would be <24 (= age/4 + 4). Note that A-a gradient may also increase by 5-7 mmHg with each 10% increase in FiO2.
Assuming an FiO2 of 0.6 with 15L/min NRB:
- pAO2 = FiO2 x (Patm – PbH20) – (PaCO2/ 0.8) = (0.6 x (760-47)) – (137/0.8) = (427.8) – ( 171.25) = 257
- paO2 = 161
- A-a gradient = pAO2 – paO2 = 257 – 161 = 96 (see A-a gradient calculator by MDCalc)
Thus, although the patient is not overtly hypoxaemic there is an increased A-a gradient (even when accounting for age and increased FiO2). This cannot be explained by hypercapnoea.
Common reasons for an increased A-a gradient in the context of decreased level of consciousness are atelectasis and aspiration. Cardiogenic or neurogenic pulmonary oedema may also occur. The patient’s previous pulmonary embolus could be contributing and the patient may have also had other pre-existing lung conditions (potentially contributing to the slow respiratory wean off tracheostomy).
Q3. What are the important causes of hypercapnia?
Causes of hypercapnia can be classified into:
- decreased removal of CO2
- decreased minute ventilation
- CNS Causes (Drugs, brainstem, central apnoea, Spinal cord injury)
- Neuromuscular (GBS, NMJ disorder e.g myaesthenia, Myopathy)
- Respiratory (reduced compliance – e.ge pulmonary oedema; reduced chest wall: kyphosis, obesity; increased airway resistance (i.e. COPD, Asthma).
- Metabolic (metabolic alkalosis, hypothyroid, hypothermia)
- increased dead space
- long ventilator tubing
- large PE
- Decreased cardiac output
- decreased minute ventilation
- increased production of CO2
- Hyperthermia
- Hyperthyroidism
- Seizures/status epilepticus
Q4. How would you approach this Code Blue (assuming no isolation precautions are required)?
ABCD approach – assess for airway patency and manage appropriately, then assess for reversible causes.
A: assess for ongoing airway patency. Things to look for include the presence of stridor, secretions and foreign bodies, snoring or neck swelling.
- Airway manouevers if required to achieve airway patency (head tilt, chin lift, jaw thrust) combined with airway adjuncts if tolerated (Guedel oropharyngeal airway (OPA) and/or nasopharyngeal airway (NPA)) and bag-valve-mask ventilation (BVM)
- If airway reflexes intact – could consider non-invasive ventilation (NIV) if conscious state improved.
Once airway patency is established, assess for immediately reversible causes such as:
- opioids (could give naloxone)
- hypoglycaemia
In this fictional case, you find that:
- SpO2 improved to 100% over 5 minutes with BVM using Guedel + NPA
- Normal blood glucose, no recent opiate use
- Level of consciousness does not improve sufficiently so the patient was intubated for airway protection rather than attempting NIV
Q5. What are the indications for CPAP/NIV? When do we use one or the other?
CPAP is continuous positive airway pressure and NIV is non-invasive ventilation. The terminology is often confusing as CPAP is also a non-invasive form of respiratory support and NIV is often referred to by the tradename BiPAP.
In general terms, CPAP is useful for improving oxygenation whereas NIV is useful for improving ventilation (CO2 clearance).
CPAP has well-defined roles in patients with acute pulmonary oedema and obstructive sleep apnoea. It is sometimes used to improve oxygenation in patients with atelectasis, especially with coexistent obesity, although definitive evidence of benefit is lacking. CPAP can also be used for pre-oxygenation prior to intubation.
NIV has welldefined roles in patients with COPD, including after extubation. It is also useful in asthma and for patients with hypercapnia due to neuromuscular disease or chest wall deformities.
The role of CPAP and NIV in acute respiratory distress syndrome and pneumonia remains controversial. Some patients may respond (e.g. over a few hours), however it is important to avoid delaying intubation when indicated. All critically ill patients receiving CPAP or NIV should be closely monitored for complications or the development of contra-indications.
In summary, the indications include:
- Hypercapnia (treated with NIV)
- acute exacerbation of chronic obstructive pulmonary disease (COPD)
- Acute asthma
- post extubation acute respiratory failure
- Obstructive Sleep Apnoea
- Cystic fibrosis
- hypercapnia due to neuromuscular disease or chest wall deformities
- Hypoxia (treated with CPAP)
- Acute Pulmonary Oedema
- Respiratory failure from rib fractures
- If not for intubation
- Preoxygenation pre intubation
| CPAP | BiPAP | |
| Settings | PEEP FiO2 | PS/PEEP FiO2 |
| Type of respiratory failure | Type 1 | Type 2 |
| PEEP | Splints airways open Prevents derecruitment Prevents atelecto/biotrauma Decreases work of breathing | |
| PS | N/A | Increases tidal volume Increases CO2 clearance Decreases work of breathing |
Q6. What are the contraindications for CPAP/NIV?
Most contra-indications are relative rather than absolute and require expert clinical judgement to weigh the risks and benefits.
Contra-indications include:
- conscious state impaired (as they may aspirate) – present in this case
- inability to protect airway (if they have a poor cough and can’t clear their secretions – these then get pushed down)
- cardiac/respiratory arrest – present in this case
- untreated pneumothorax
- marked haemodynamic instability
- Following UGI surgery (to be discussed with the surgical team prior)
- MaxFax surgery
- Base of skull fracture (risk of pneumocephalus)
- Intractable vomiting (aspiration risk)
Based on these contraindications, the decision to intubate the patient was correct.
References
- Deranged Physiology. 2015. Approach to hypercapneic respiratory failure. https://derangedphysiology.com/main/required-reading/respiratory-medicine-and-ventilation/Chapter%208.1.1/approach-hypercapneic-respiratory-failure
- West, JB. Respiratory Physiology. Ninth Edition. Wolters Kluwer Health/Lippincott Williams & Wilkins, Philadelphia, 2012.
- Deranged Physiology. 2018. Non-invasive mechanical ventilation. https://derangedphysiology.com/main/required-reading/respiratory-medicine-and-ventilation/Chapter%202.5.6/non-invasive-mechanical-ventilation
- Lumb, A. Nunn’s Applied Respiratory Physiology. Elsevier, 2017.
All case-based scenarios on INTENSIVE are fictional. They may include realistic non-identifiable clinical data and are derived from learning points taken from clinical practice. Clinical details are not those of any particular person; they are created to add educational value to the scenarios.