Author: Diarmuid O’Briain
Peer reviewers: Paul Nixon, Chris Nickson
Everything ECMO 031
A 40-year-old male with cardiogenic shock due to dilated cardiomyopathy has been cannulated to allow commencement of V-A ECMO to provide circulatory support.
A distal perfusion cannula (DPC) is inserted as part of your unit’s routine V-A ECMO initiation procedure.
Q1. What are the vascular complications of peripheral V-A ECMO?
- Vessel perforation with subsequent haemorrhage
- Arterial dissection
- Limb ischaemia, occasionally requiring progressing to compartment syndrome, requiring fasciotomy, or even amputation.
- Pseudoaneurysm formation
- Incorrect placement of cannula at the time of insertion, eg venous cannula inserted into an artery
- Acute embolism (e.g. air or thrombus)
Q2. How common is limb ischaemia in peripheral V-A ECMO?
A recent meta-analysis of 1886 patients reported a 17% incidence overall of limb ischaemia, with 10% progressing to compartment syndrome and requiring fasciotomy, and 4.7% needing amputation1.
Q3. What is the mechanism of limb ischaemia in peripheral V-A ECMO?
A number of mechanisms have been described…
- Obstruction to arterial flow
- Loss of pulsatile flow
- Stasis and venous congestion in the femoral vein due to wide bore access cannulation occluding venous return
- Systemic vasoconstriction from shock, or pharmacological vasoconstriction.
Q4. What options exist for distal arterial flow?
There are several techniques used to improve distal arterial flow and improve leg ischaemia.
The commonest options are:
- Insertion of a distal perfusion cannula (DPC) in a site distal to the arterial return cannula (See image and description below)
- Formation of a T- graft or an end-to-side chimney graft, in place of direct arterial cannulation. These options employ a surgical technique and are rarely used at the Alfred. A complication of these techniques is uncontrolled distal blood flow leading to hyperaemia of the limb. This can be mitigated with the application of an arterial band distal to the anastomosis point of the vessel.
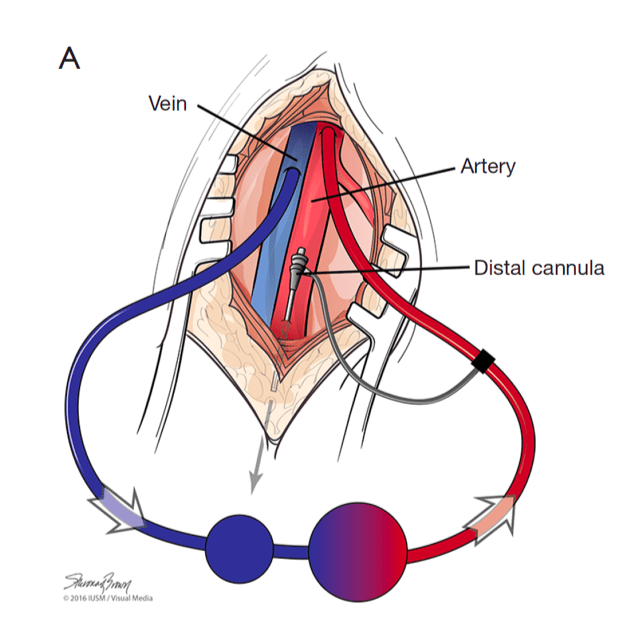
T-graft

Image source: Makdisi G, Makdisi T, Wang IW. Use of distal perfusion in peripheral extracorporeal membrane oxygenation. Ann Transl Med 2017;5(5):103. Available at URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5360617/ (Click image for source)
End-to-side chimney graft
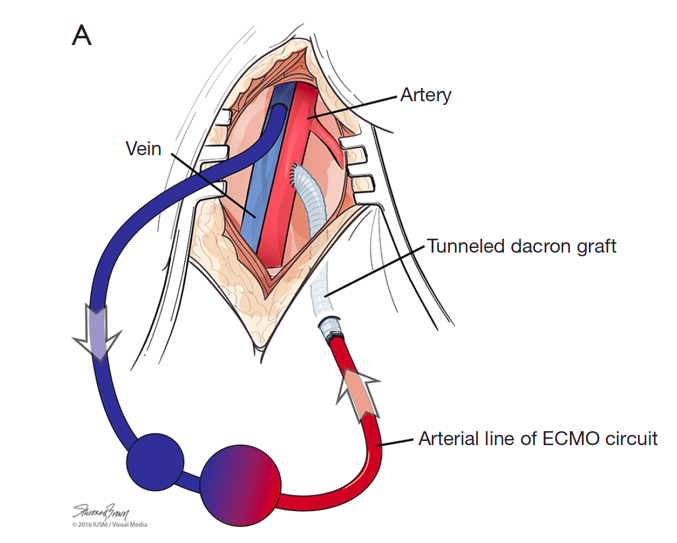
Image source: Makdisi G, Makdisi T, Wang IW. Use of distal perfusion in peripheral extracorporeal membrane oxygenation. Ann Transl Med 2017;5(5):103. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5360617/ (Click image for source)
Q5. What is a distal perfusion cannula (DPC)? How does it work?
A DPC is a small cannula which is placed in the superficial femoral artery distal to the insertion point of the femoral return cannula. In our institution, we use either 6, 8 or 9 French devices. They are used to ensure adequate arterial flow to the distal leg, to prevent limb ischaemia when peripheral V-A ECMO is in use. It is connected to the arterial return cannula and diverts a portion of this blood distally. The relative small size of the DPC compared to the arterial return cannula limits flow and thus hyperaemia of the limb.
Q6. Do all ECMO units routinely use distal perfusion cannulas (DPCs)?
No.
Some units, such as the Alfred, routinely use DPCs in all VA-ECMO patients as prophylaxis against limb ischaemia. Whereas some units use them as a rescue technique when limb ischaemia occurs. The majority of studies demonstrate a lower incidence of limb ischaemia when a distal perfusion cannula is used, compared to when one is not inserted2.
Q7. What are the complications of distal perfusion cannula insertion?
It is hard to accurately assess the complications directly related to the insertion of a distal perfusion cannula, compared to the insertion of the larger return cannula in the femoral artery.
Studies have noted ischaemic complications such as
- Superficial femoral artery thrombosis
- Compartment syndrome
- Necrotic toes
- Peripheral neuropathy
- Requirement of embolectomy
- Requirement of fasciotomy
- Requirement of amputation.
Again however, it is difficult to determine whether these complications were due to the distal perfusion cannula itself, or due to the primary femoral return cannulation. Local complications can also occur, such as
- Femoral artery dissection
- Pseudoaneurysm formation
- Groin haematoma
- Femoral artery laceration.
One complication of a DPC is the inability to control the amount of blood flow down the leg which can result hyperperfusion of that distal limb3, however as mentioned above the relatively small size of the DPC usually limits this phenomenon.
Q8. Where is the distal perfusion cannula inserted?
At the Alfred they are always inserted into the superficial femoral artery (SFA) just distal to the insertion point of the femoral return cannula. This provides the leg with anterograde blood flow. However, insertion points have also been described in the posterior tibial artery4 and dorsalis pedis5, which perfuse the leg with retrograde flow.
Q9. Does leg ischaemia correlate with V-A ECMO outcomes?
Papers have suggested that leg ischaemia does not correlate with ECMO outcomes6. However, more recent papers suggested that leg ischaemia is an independent risk factor for in-hospital death7. Vascular complications have also been shown to be associated with unsuccessful weaning of ECMO8. Obviously, leg amputation would have a significant effect on quality of life.
Q10. When should a distal perfusion cannula be inserted?
At the Alfred, we find the best time to insert a distal perfusion cannula is during ECMO insertion. We find it easiest to insert the DPC wire first, then cannulate the artery and vein percutaneously, and lastly insert the distal perfusion cannula over the wire in situ. The superficial femoral artery is a small vessel, therefore insertion of the wire prior to ECMO cannulation significantly improves ultrasound visibility and success. This method is impractical during ECMO CPR cases, and in these cases we insert the distal perfusion cannula after initiation of ECMO once the patient is stabilised.
Q11. How is a distal perfusion cannula (DPC) inserted into the superficial femoral artery (SFA)?
The DPC is inserted using a Seldinger technique of catheter over wire. We use ultrasound guidance for all insertions and employ a 2-person method. Operator 1 cannulates the SFA using in-plane imaging and Operator 2 feeds the wire down the artery under ultrasound guidance once arterial puncture occurs. As the vessel is small and often deep, we find this 2-person technique limits needle movement and allows visualization of the wire progressing down the distal vessel.
In the video below, you see the needle puncture the superficial femoral artery. You can see the common femoral artery on the right side of the screen branching into the SFA (upper) and profunda femoris (lower). The superficial femoral artery is critical in supplying oxygenated blood to the lower limb. The profunda femoris is also called the deep femoral artery (DFA) or deep artery of thigh.

Ultrasound image showing SFA cannulation and guidewire insertion to facilitate insertion of a DPC.
References
- Cheng R, Hachamovitch R, Kittleson M, et al. Complications of extracorporeal membrane oxygenation for treatment of cardiogenic shock and cardiac arrest: a metaanalysis of 1,866 adult patients. Ann Thorac Surg 2014;97:610–6. [pubmed] [article]
- Juo, Y. , Skancke, M. , Sanaiha, Y. , et al. (2017), Efficacy of Distal Perfusion Cannulae in Preventing Limb Ischemia During Extracorporeal Membrane Oxygenation: A Systematic Review and Meta‐Analysis. Artificial Organs, 41: E263-E273. [pubmed]
- Belle L, Mangin L, Bonnet H, et al. Emergency extracorporeal membrane oxygenation in a hospital without on-site cardiac surgical facilities. EuroIntervention 2012;8:375-82. [pubmed]
- Spurlock DJ, Toomasian JM, Romano MA, et al. A simple technique to prevent limb ischemia during veno-arterial ECMO using the femoral artery: the posterior tibial approach. Perfusion 2012;27:141-5. [pubmed]
- Kimura N, Kawahito K, Ito S, Murata S, et al. Perfusion through the dorsalis pedis artery for acute limb ischemia secondary to an occlusive arterial cannula during percutaneous cardiopulmonary support. J Artif Organs 2005;8:206-9. [pubmed]
- Bisdas T, Beutel G, Warnecke G, et al. Vascular complications in patients undergoing femoral cannulation for extracorporeal membrane oxygenation support. Ann Thorac Surg 2011;92:626-31. [pubmed] [article]
- Hei F, Lou S, Li J, et al. Five-year results of 121 consecutive patients treated with extracorporeal membrane oxygenation at Fu Wai Hospital. Artif Organs 2011;35:572-8. [pubmed]
- Aubron C, Cheng AC, Pilcher D, et al. Factors associated with outcomes of patients on extracorporeal membrane oxygenation support: a 5-year cohort study. Crit Care 2013;17:R73. [pubmed] [article]
All case-based scenarios on INTENSIVE are fictional. They may include realistic non-identifiable clinical data and are derived from learning points taken from clinical practice. Clinical details are not those of any particular person; they are created to add educational value to the scenarios.