Author: Jonah Powell-Tuck
Reviewers: Aidan Burrell, Chris Nickson
Everything ECMO 011
An obese 49-year-old man is being treated in the ICU for severe bilateral pneumonia. He has been on femoral-femoral VV ECMO for the past 24 hours. The pump blood flow is 5L/min and fresh gas flow is 8L/min. He is ventilated with SIMV PC 15 cmH20 PEEP 15 cmH20 with unchanged tidal volumes of 20ml. His observations are HR 125/min, BP 90/50 mmHg on 0.6mcg/kg/min noradrenaline with oxygen saturations (SpO2) of 82%.
Q1. What initial ECMO circuit checks should be made?
Ensure adequate ECMO circuit oxygenation:
- Is the oxygen attached and running?
- Is the ECMO gas flow FiO2 1.0?
Ensure pump flow calibrations are correct:
- Recalibrate the ECMO pump flow as described in Everything ECMO case 001 Q1
Ensure there is no recirculation:
- Are the access and return cannula tips 10cm apart?
- Is colour difference seen between access and return lines?
- Is the pre-oxygenator oxygen saturation appropriate?
Ensure the oxygenator is functioning adequately:
- Check post-oxygenator gases
Pre-oxygenator saturations are 50%. Post oxygenator pO2 is 450mmHg. An echocardiogram is performed revealing a hyperdynamic LV with mildly impaired RV. The estimated cardiac output is 10L/min. The return cannula is seen in the RA with the tip of the access cannula 10cm lower in the IVC.
Q2. Why is the patient still hypoxaemic?
The patient is hypoxaemic as too much of the patient’s deoxygenated blood is shunting past the ECMO circuit, rather than flowing through the ECMO circuit (see Figure 1)
- The patient’s overall oxygenation results from contributions from both the ECMO circuit and the lungs. If the lungs are having a negligible effect on oxygenation the patient is almost entirely reliant upon the ECMO circuit for gas exchange.
- 5L of blood per minute is passing through the ECMO circuit and is returned to the circulation fully saturated. However, the patient’s cardiac output is 10L/min, meaning 5L (50%) of blood bypasses the ECMO circuit. The lungs are failing and therefore have impaired ability to oxygenate blood, meaning 50% of cardiac output passes through to systemic circulation with minimal oxygenation.
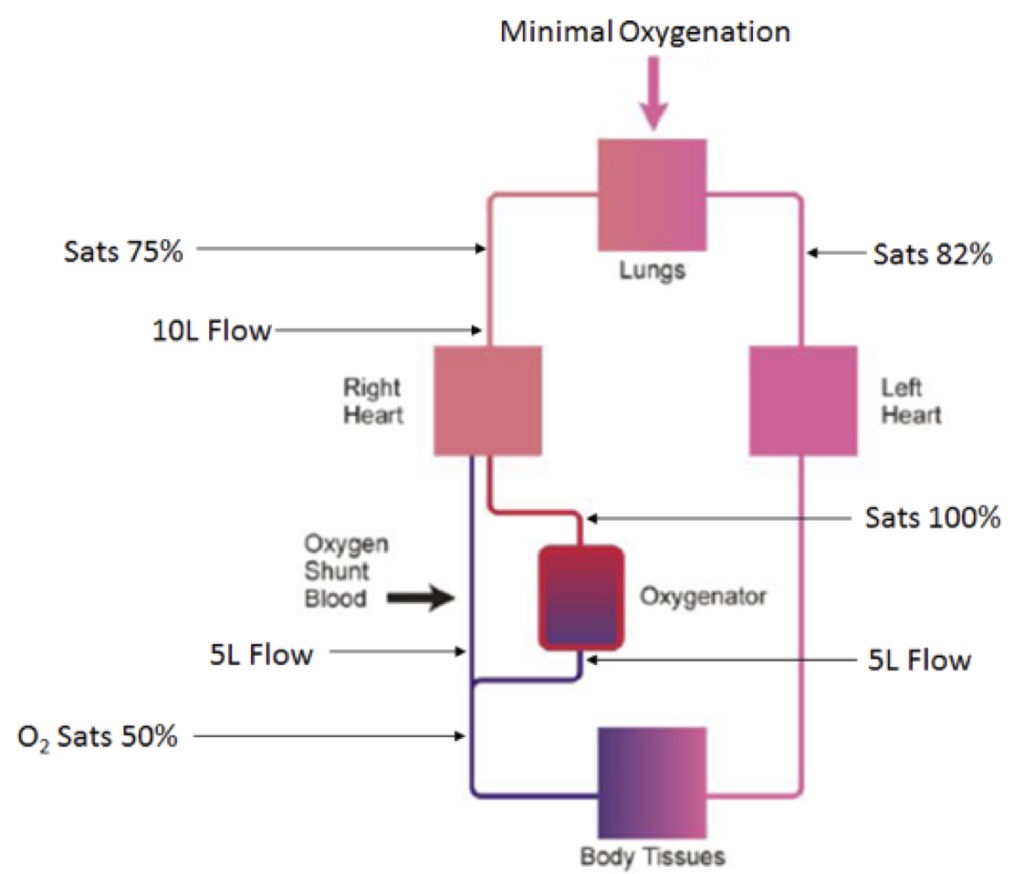
Figure 1. Schematic diagram describing the degree of blood oxygenation at different site’s of the patient’s circulation while on VV ECMO.
Q3. Increasing the pump speed leads to ‘kicking’ of the access line with no increase in pump flow. What are the options at this stage?
Increasing the pump speed is causing access insufficiency (see Everything ECMO 003). After decreasing the pump speed until the ‘kicking’ resolves, the options include:
- Improving the patient’s oxygenation by adjusting mechanical ventilation
There appears to be little recruitability in this patient and little room to move whilst maintaining lung protective ventilation. More aggressive ventilation is likely to result in worsening ventilator-induced lung injury. Treatment should focus on the patient’s underlying condition. A negative fluid balance should be targeted however access insufficiency and high noradrenaline requirements may limit this. Consideration should be given to nitric oxide therapy, proning and alternative ventilation strategies such as APRV, however, none of these measures are likely to lead to rapid improvements in the patient’s condition. - Reduce the patient’s oxygen demand
Increasing sedation, paralysing and cooling the patient will help to reduce the patient’s oxygen demand. These interventions may also allow pump flow to be increased. However, they are associated with side effects such as weakness, delirium and increased risk of infection. - Reduce the patient’s cardiac output
β-blockade could be used to reduce the patient’s cardiac output thereby reducing the proportion of “shunted” non-oxygenated blood reaching the arterial system. However, this may not be advisable in this case as the patient is hypotensive on high dose vasopressors. The role of β -blockers in sepsis is controversial (see the LITFL CCC entry on Catecholamines, Beta-blockade and Critical Illness). - Accept low oxygen saturations
It is unclear what degree of hypoxia is acceptable in such patients, the ARDSnet trial targeted pO2 55-88mmHg and Sats 88-95%. Subnormal oxygen saturation targets in ARDS patients is associated with long-term cognitive impairment (see LITFL CCC entry on Oxygen Saturation Targets in Critical Illness). Importantly, a high cardiac output with lower saturations may result in a higher DO2 than a lower cardiac output with improved oxygen saturations. - Increase pump flow and give fluid for access insufficiency
You are unlikely to achieve the circuit blood flows necessary to overcome the shunt physiology created by the high cardiac output using this approach. Very positive fluid balances lead to further lung injury impairment of gas exchange and they are also associated with higher mortality in severe ARDS (see LITFL CCC entry on De-resuscitation and Positive Fluid Balance) - Switch to a high flow ECMO circuit configuration
Inserting a second access cannula will allow high pump flows whilst avoiding the problem of access insufficiency. Oxygenation is improved without the need to decrease cardiac output.
Q4. What are indications for high flow ECMO circuit configurations?
Key indications are:
- VV ECMO with access insufficiency
- VV ECMO with refractory hypoxia
- Peripheral VA ECMO with severe differential hypoxia

Figure 2. VV ECMO High Flow Configuration (left) and VA ECMO High Flow Configuration (right)
Q5. How will switching to a high flow ECMO circuit configuration improve this patient’s oxygenation?
Two access cannulas at two different anatomical sites will allow higher flows to run through the ECMO circuit whilst avoiding the problem of access insufficiency
- The higher flows will increase the proportion of the blood that is oxygenated via the ECMO circuit thereby reducing the “shunted” portion, consequently improving arterial oxygen saturation
Q6. How is the conversion to a high-flow configuration made?
These are the steps involved:
- Exclude left internal jugular occlusion on ultrasound
- Oxygenate the patient with 100% O2 using the ventilator
- Insert a short “arterial” cannula into right internal jugular (RIJ) vein using aseptic technique
- Drape and clean the femoral access cannula with iodine-based antiseptic
- Prime a Y connector attached to long extension tubing with saline and attach the long extension to the RIJ cannula using the ‘underwater seal’ technique
- Clamp the circuit and the femoral access cannula then remove the access tubing from the femoral cannula
- Attach the femoral cannula to the short end of the y connector
- Attach the access tubing to the y connector using the ‘underwater seal’ technique
- After ensuring there is no air in the circuit remove clamps and re-establish pump flow
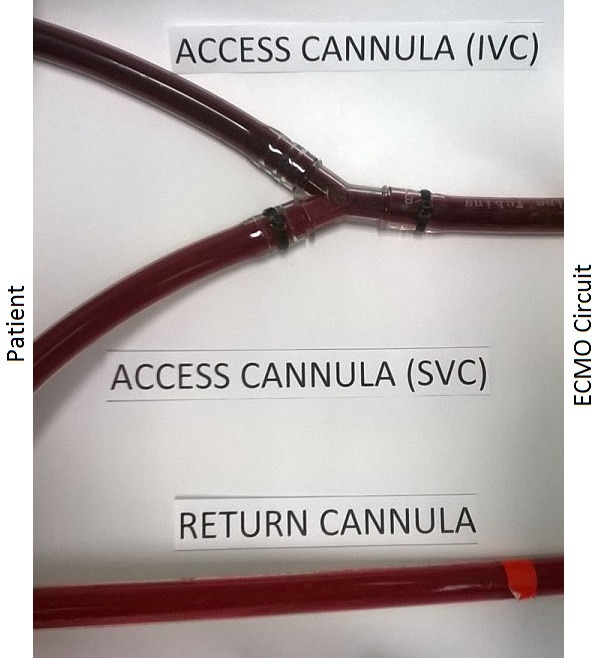
Figure 3. Photograph of y connector in high flow circuit. The patient is out of shot on the left and the ECMO circuit is on the right.
Q7. Explain the difference in colour between the blood from the IVC access cannula and the SVC access cannula in Figure 3.
The blood from the SVC access cannula appears lighter as the oxygen saturation is greater than the blood entering the IVC access cannula.
- This is a result of recirculation of oxygenated blood from the return cannula.
- The relative positions of the three cannulae seen in figure 4 demonstrate how the return cannula may direct oxygenated blood toward the SVC, leading to the SVC cannula having a greater degree of recirculation than the IVC cannula.
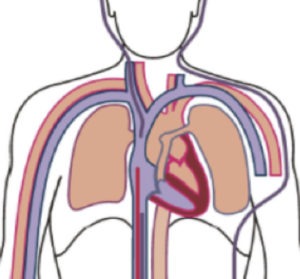
Figure 4. Position of Cannulae in High Flow VV ECMO
References and Links
- Pellegrino V, Murphy D, Hockings L, Roberts L. Extracorporeal Membrane Oxygenation (ECMO). Alfred ICU, 2012
What about the target Hb level
I did dual VV drainage for 35 y female with BMI of 75 as it was very difficult to maintain Oxygen sat above 80 withe one drainage cannula esp.with starting CRRT for AKI and strategy to dry the native lung.I inserted Rt int J. 21 cannula and coonected through Y shaped 3/8 with lt femoral 25 cannula and returning was 23 Rt fem. After change configuration we achived sat above 93%
Pingback: LITFL Review 282 • Life in the Fast Lane • LITFL • Medical Blog