Author: Aidan Burrell
Reviewers: Vinodh Nanjayya, Chris Nickson
Everything ECMO 013
A patient who was previously supported with VA ECMO has just returned from the operating theatre following the insertion of a left ventricular assist device (VAD).
Q1. What are the 4 key components of a VAD?
The 4 key components (see Figure 1) are:
- Inflow cannula – Implanted (through a cored out hole) into the apex of the left ventricle
- Blood pump – Has a moving rotor that spins with either an axial or centrifugal mechanism. The pump sits in the ‘pump pocket’, which may be intra-pericardial (e.g. HeatWare and HeartMate 3) or pre-peritoneal (e.g. HeartMate 2)
- Outflow cannula – Connects the pump to the ascending aorta via a Dacron graft.
- Percutaneous lead (or drive line) – Travels from pump through abdominal wall to the outside controllers, and contains cables for power and data.

Figure 1. VAD components
Q2. What are the differences between axial pumps and centrifugal pumps?
Centrifugal pumps (Eg: HeartWare, HeartMate III) have a rotor or impeller that is magnetically levitated (elevated) in the pump housing (see Figure 2). Current passing through the magnets on the impeller causes it to spin. Blood enters the center of the impeller under negative pressure. The rotating vanes then accelerate the blood from the centre to the outside of the impeller. This blood is under positive pressure, and drains out of the pump via the discharge nozzle into the outflow cannula.

Figure 2. Centrifugal pump (from Aaronson et al, 2012)
Axial pumps (eg: HeartMate II) use a turbine or corkscrew mechanism to propel blood in a path parallel to the rotor into the outflow cannula. These spin at higher RPMs than centrifugal pumps, have higher power requirements, and may be associated with more blood trauma and coagulopathy.
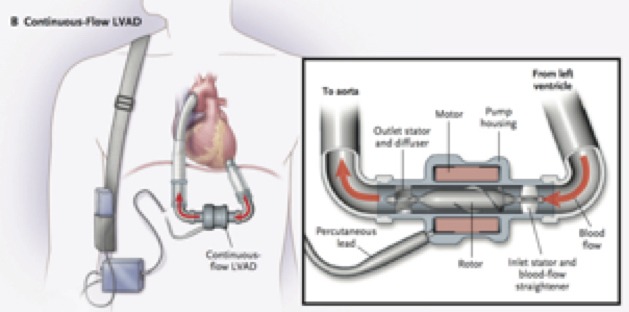
Figure 3. Axial pump (from Slaughter et al, 2009)
Q3.Your hospital stocks 3 types of LVAD: the HeartMate II, the HeartWare, and the HeatMate III. Compare and contrast these common types of LVAD.
This table provides a useful comparison:
| HeartMate II | Heart Ware | Heart Mate III | |
| Mechanism | Axial | Centrifugal | Centrifugal |
| Flow | Continuous | Continuous | “Pulsatile” pump speeds up and slows every 2 seconds (30 bpm) |
| Insertion | Pump pocket | Pericardial space | Pericardial space |
| Pilot trial date | 2003 | 2009 | 2014 |
| Speed, rpm | 8600-9800 | 2500-2900 | 5000-6000 |
| Power, watts | 6-7 | 3-7 | 3-7 |
| Flow, L/min | 4-8 | 4-7 | 3-6 |
Q4. The patient’s arterial line has a flat trace with a mean arterial pressure (MAP) of 65mmHg. What can you tell about the circulation from the arterial line of a LVAD patient?
Patients with LVAD have dual circulations.
The native cardiac circulation is responsible for the pulsatility on the arterial trace, while the LVAD is responsible for continuous flow component (which is manifested as a MAP).
The relative proportion of each of these systems can be inferred from the degree of pulsatiliy on the arterial trace. The flat arterial trace in this patient suggests most of the circulation is going through the LVAD, and the native circulation is contributing very little. This can occur early postoperatively in patients with severe heart failure.
The graph of VAD flow rate versus rotational speed for the HeartWare VAD (see Figure 4) shows that as the LVAD rotational speed increases, the VAD flow rate increases and the pulsatility decreases. If the speed is increased too high, negative inflections in flow result which indicate ‘suck down’ events.
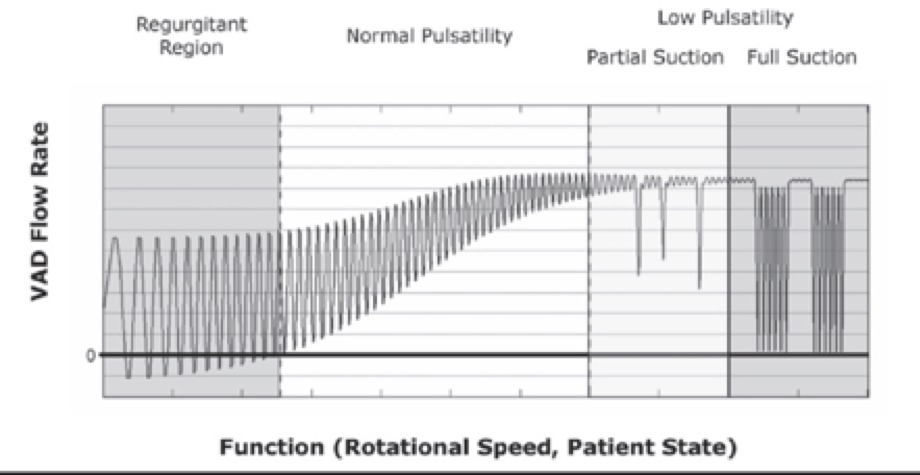
Figure 4. VAD flow rate versus pump speed (rpm) for the HeartWare VAD device.
Q5. The patient’s lactate starts to increase and the LVAD flow fall to 3L/min. The MAP is 90 mmHg. Is administering an intravenous fluid bolus the answer? Why?
No. Unless there is suck down, administering more IV fluid will have minimal impact on the VAD output.
LVAD flow is dependent on the preload, but in a slightly different way to the native heart. In Figure 5, the graph on the left shows the Frank-Starling curve displaying the normal relationship between preload and cardiac pump output (or stroke volume in this case). As you increase preload (i.e., fluid bolus), cardiac output increases up until a point where it has little effect.
The equivalent relationship for a VAD pump is shown in Figure 5 by the graph on the right: the curve describing the relationship between preload and output is “flatter”. This implies that an increase in preload does not result in much of an increase in LVAD output. Furthermore, at low preload the pump doesn’t have any output at all – and ‘suck down’ occurs. Hence, the VAD is preload dependent – it needs some preload to work, but increasing the preload further doesn’t have much impact on pump output.
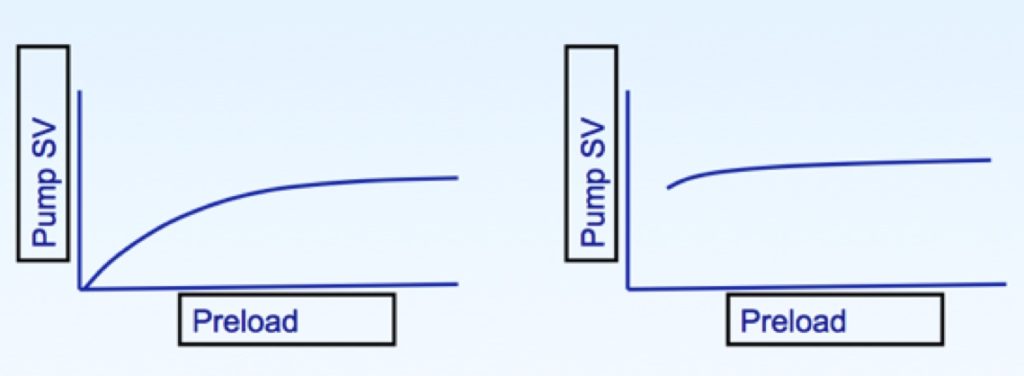
Figure 5. Stroke volume versus preload for the native heart (left) and an LVAD (right).
Q6. The patient’s LVAD flow remains low. Is there anything else you can do to improve LVAD flow?
The LVAD pump flow is inversely proportional to the pressure differential across the pump head (shown in Figure 6). This pressure differential, is essentially the difference between the pre-load and afterload. As the afterload (represented by MAP) increases, the pressure differential across the pump head increases leading to a reduction in pump flow. Similarly, as the afterload decreases, the pressure differential decreases and the pump flow increases. Unlike preload, the relationship between afterload and LVAD flow is more dynamic in the physiological ranges that are used in patients. Hence, a drop in MAP from 85 mmHg to 65 mmHg results in a large increase in VAD pump flow rate from 1.5 L/min to 5 L/min.
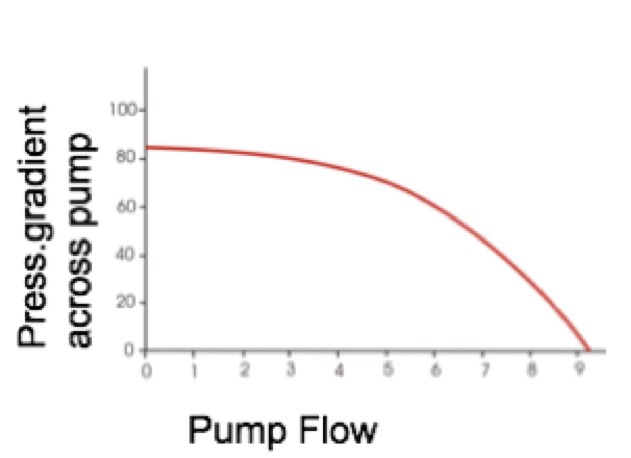
Figure 6. Pressure gradient across pump versus pump flow for an LVAD
Knowing that VADs are ‘preload dependent but afterload sensitive’, you start a sodium nitroprusside infusion and lower the MAP to 65 mmHg. The VAD output increases by 2.5 L/min.
Q7. The patient stabilises after your intervention. You recall that when the perfusionist handed over the patient, she mentioned the VAD was set at 2600 rpm, but above this they had “suck down”. Currently the LVAD output is 5.5 L/min. How do you set the LVAD speed?
Setting the LVAD speed is optimally done while performing an echocardiogram. The aim is to have the inflow cannula sitting in the middle of the left ventricle with the ventricle decompressed but not sucking down.
If the LVAD speed is too high the LV internal cavity size will shrink down resulting in ‘suck down’. Suck down occurs when the ventricular endocardial border is sucked into inflow cannula. Although a pump speed that is set too high is the most common cause, it can also result if the inflow cannula is malpositioned. Suck down causes intermittent drops in flow, damage to the ventricle, haemolysis and ventricular arrhythmias.
On the other hand, if the LVAD speed is too low, the ventricle will remain overloaded and the patient may have persisting heart failure.
In practice, this usually involves turning the speed up then down to find the ‘sweet spot’ between the above two problems.
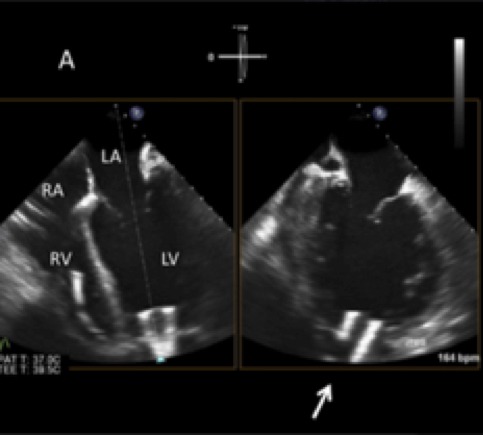
Figure 7. Transoesophageal echocardiogram showing the inflow cannula sitting in the apex and sitting in the centre of the left ventricular internal cavity (from Stainback et al, 2013)
Q8. Later on that evening, the patient suddenly goes into cardiac arrest. Should you start CPR?
LVAD patients differ from other cardiac arrest patients as the pump will still provide some ongoing flow. In general, the manufacturers recommend you to lower the speed of the LVAD (HeartMate II 8000 rpm, HeartWare 1800 rpm and Heartmate III 4000 rpm) as this reduces the risk of suck down and loss of forward flow through the pump.
The decision to start CPR depends on your local guidelines and will depend on the cause of the cardiac arrest. The risk of starting CPR is that it may not contribute much to flow and may damage the pump. On the other hand, CPR may be appropriate for a patient with complete pump failure.
References
- Aaronson KD, Slaughter MS, Miller LW, et al. Use of an intrapericardial, continuous-flow, centrifugal pump in patients awaiting heart transplantation. Circulation. 2012; 125(25):3191-200. [pubmed]
- Slaughter MS, Rogers JG, Milano CA. Advanced heart failure treated with continuous-flow left ventricular assist device. The New England journal of medicine. 2009; 361(23):2241-51. [pubmed]
- Stainback RF, Estep JD, Agler DA, et al. Echocardiography in the Management of Patients with Left Ventricular Assist Devices: Recommendations from the American Society of Echocardiography. Journal of the American Society of Echocardiography : official publication of the American Society of Echocardiography. 2015; 28(8):853-909. [pubmed]
Excellent and lucid