Labs and Lytes 020
Author: Chris Nickson
Reviewer: David Pilcher
You are asked to review a 50 year-old woman with worsening hypoxaemia during the night shift in ICU. Earlier that day she was admitted from the operating theatre following bilateral lung transplantation for idiopathic pulmonary fibrosis.
Q1. What is your differential diagnosis for hypoxaemia in this patient?
As with any intubated patient, hypoxaemia can be due to “DOPES”:
- displaced endotracheal tube
- obstructed endotracheal tube
- patient factors (especially pulmonary disorders)
- equipment factors (e.g. oxygen failure)
- “stacked breaths” (dynamic hyperinflation)
In this case, the endotracheal tube is correctly positioned and a suction catheter passes freely. You review the ventilator and see that it is working perfectly.
This leaves consideration of the common (and commonly dangerous) causes of hypoxaemia post-lung transplant:
- primary graft dysfunction
- collapse (e.g. lobar collapse due to post-operative blood clot or sputum)
- pneumonia and other infective causes
- hyperacute rejection (rare, due to a sensitised recipient producing antibodies that target the lung allograft)
- cardiogenic pulmonary edema
- pulmonary vascular disease
- pulmonary embolus
- pulmonary venous obstruction (e.g. kinking or thrombosis)
- pleural disorders
- pneumothorax (e.g. air leak from anastomosis dehiscence)
- chylothorax (e.g. surgical injury to lymphatics)
- hemothorax (e.g. in the context of a blocked ICC)
Note also that, in patients with single lung transplants, dynamic hyperinflation can be a particular issue. This occurs because the allograft has relatively poor compliance so the native lung preferentially hyperexpands at high pressure settings. However, significanct dynamic hyperinflation is highly unlikely in bilateral lung transplant patients (unless the donor had asthma…!).
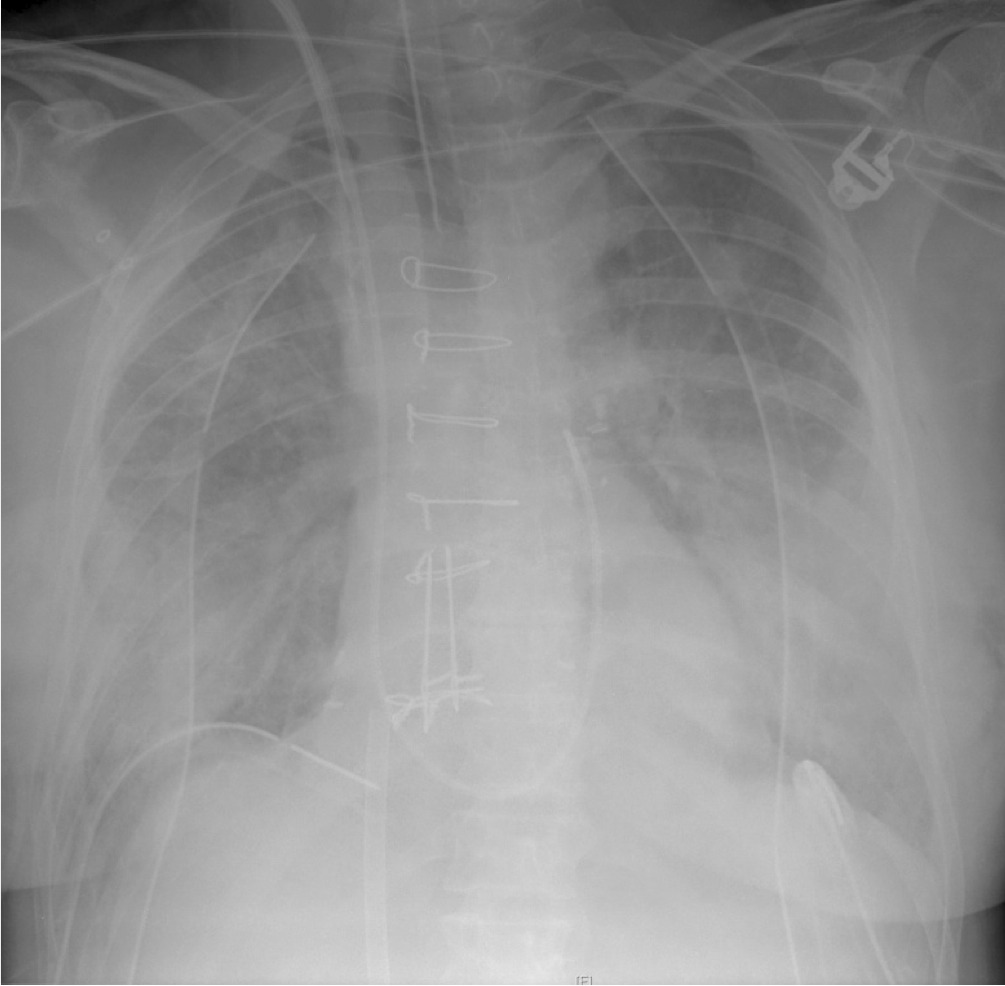
Q2. What does the CXR in Q1 show, and what diagnosis do you suspect?
The notable finding is the presence of diffuse bilateral lung opacities. This is most likely due to primary graft dysfunction (PGD).
PGD affects up to 30% of lung transplant patients in some series and is the most frequent and significant cause of early mortality following lung transplantation. In addition to bilateral infiltrates and a decreased PF ratio (PaO2/ FiO2), the diagnosis of PGD requires the exclusion of cardiogenic, infective and immunological causes.
Other findings on the chest radiograph are:
- sternotomy wires
- correctly positioned pulmonary artery catheter
- right internal jugular central venous catheter
- right and left-sided apical and basal intercostal catheters
- an access cannula for V-V ECMO in the IVC (the return cannula is sited below the level of the chest x-ray)
- leads for cardiac monitoring
- no nasogastric tube!
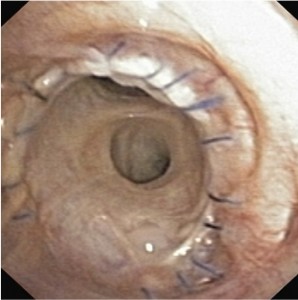
Early bronchoscopy should be performed once the patient is sufficiently stable to check that the bronchial anastomoses are healing well, obtain samples for microscopy, culture and PCR and exclude any causes of collapse such as blood clots or sputum plugs.
This is how an intact, normally healing anastomosis should look on day 1 post-lung transplant:
Pulmonary venous obstruction due to kinking, inadvertent ligation, or thrombosis can also cause pulmonary edema and (often) hypotension due to impaired flow to the left atrium. This can is best diagnosed by trans-oesophageal echocardiography.
ECMO, as demonstrated in the chest radiograph, can be used as a rescue therapy for severe primary graft dysfunction (see Q4 and Q5).
Q3. What are the underlying causes and risk factors for the most likey diagnosis mentioned in Q2?
The underlying cause of primary graft dysfunction is multifactoral. These factors include:
- the absence of lymphatic drainage from the lung allografts, so that any accumulating fluid is not effectively removed
- inflammation from pre-existing disease or surgical trauma during procurement
- poor preservation leading to ischaemia/ reperfusion injury
- ventilator induced lung injury
Numerous risk factors have been described, important ones that we consider are:
- a low PF ratio in donor
- venous thromboembolism
- blood product administration
- prolonged ischaemia times
- requirement for cardiopulmonary bypass during lung transplantation
Q4. What is appropriate management of this condition?
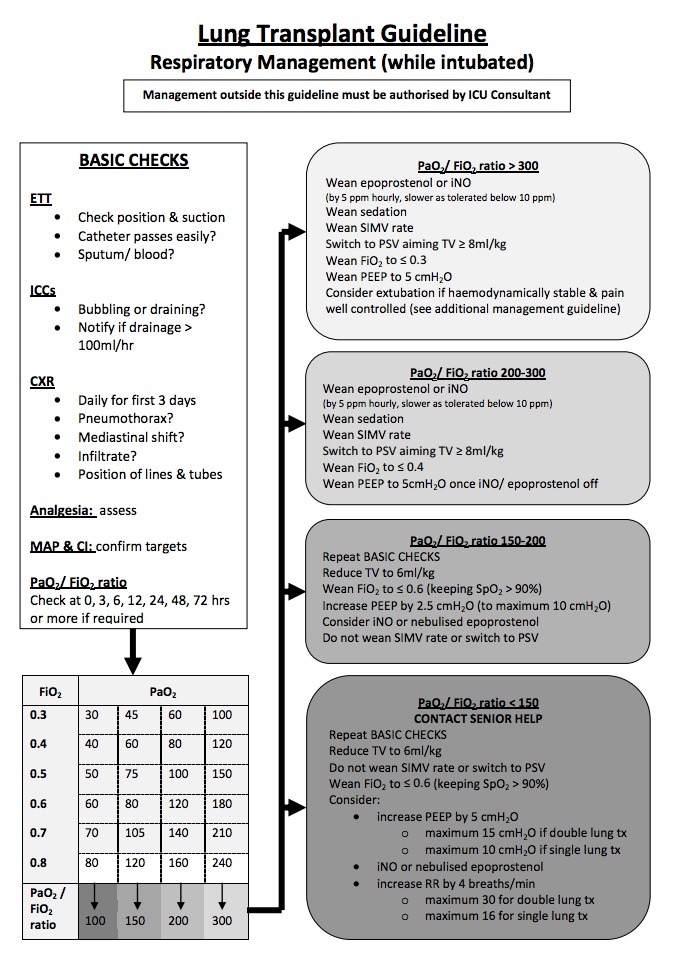
The respiratory management component of the lung transplant protocol at The Alfred ICU is primarily designed to prevent and amelioriate primary graft dysfunction. The protocol is based on regular assessment of the PF ratio as described by Currey et al, 2010.
Management of primary graft dysfunction is primarily supportive, and includes:
- exclusion of other causes of decreased PF ratio with bilateral infiltrates (e.g. infection, rejection, cardiogenic pulmonary oedema)
- avoidance of excessive fluid administration
- use of diuretics (to reduce oedema)
- protective lung ventilation (to reduce ventilator induced lung injury)
- use of PEEP to improve oxygenation (maximum PEEP of 15 cmH20 for bilateral lung transplants, maximum PEEP of 10 cmH20 for single lung transplants due to risk of dynamic hyperinflation)
- use of pulmonary vasodilators (inhaled nitric oxide, epoprostenol) to improve oxygenation and decrease pulmonary artery pressures (thus lowering the hydrostatic pressure driving alveolar capillary leak and decreasing right ventricular afterload)
- ECMO may be used as a rescue therapy allowing ‘bridge to recovery’ or, in rare refractory cases, bridge to re-transplantation.
Q5. What are the important considerations if ECMO is used as a rescue therapy in this context?
V-V ECMO can be used to provide ventilatory support. However, VA-ECMO may be used if there is significant co-existent cardiac dysfunction or pre-emptively in patients who had severe pulmonary hypertension… which has important ramifications for the lung transplant patient.
If the V-A ECMO blood flow is too high the pulmonary circulation may be bypassed. This can be problematic as the bronchial arteries are typically sacrificed during lung transplantation and there is no longer any systemic blood supply to the lungs. Transplanted lungs are dependent on the pulmonary circulation for blood supply and this must be maintained.
Thus VA-ECMO can be too much of a good thing, it avoids pulmonary congestion (especially in patients who previously had pulmonary hypertension) but may lead to lung ischaemia if too much blood flow is diverted from the transplanted lungs.
Q6. What are the prognostic implications of this diagnosis?
Primary graft dysfunction is associated with:
- prolonged mechanical ventilation
- prolonged ICU length of stay
- worse functional outcomes
- increased rates of bronchiolitis obliterans syndrome (BOS), which is chronic airflow limitation resulting from chronic allograft rejection
References and Links
- Currey J, Pilcher DV, Davies A, Scheinkestel C, Botti M, Bailey M, Snell G. Implementation of a management guideline aimed at minimizing the severity of primary graft dysfunction after lung transplant. J Thorac Cardiovasc Surg. 2010 Jan;139(1):154-61. doi: 10.1016/j.jtcvs.2009.08.031. Epub 2009 Nov 11. PMID: 19909995.
- INTENSIVE — Transplant Patients in ICU SDL
- Mishra A, Reed RM, Eberlein M. Severe, Rapidly Reversible Hypoxemia in the Early Period after Bilateral Lung Transplantation. Ann Am Thorac Soc. 2016;13(6):979-85. PMID: 27295160 [article]
thats a fun case. whats the volume of lung transplant cases you guys do?
Currently, there are about 85 lung transplant cases per year at The Alfred – a predicted 90+ cases in 2016 I think.
An interesting stat, about 1in 30 of all lung transplants that have ever occurred worldwide have taken place at The Alfred!
Cheers
Chris
Pingback: LITFL Review 158 • LITFL Medical Blog • FOAMed Review